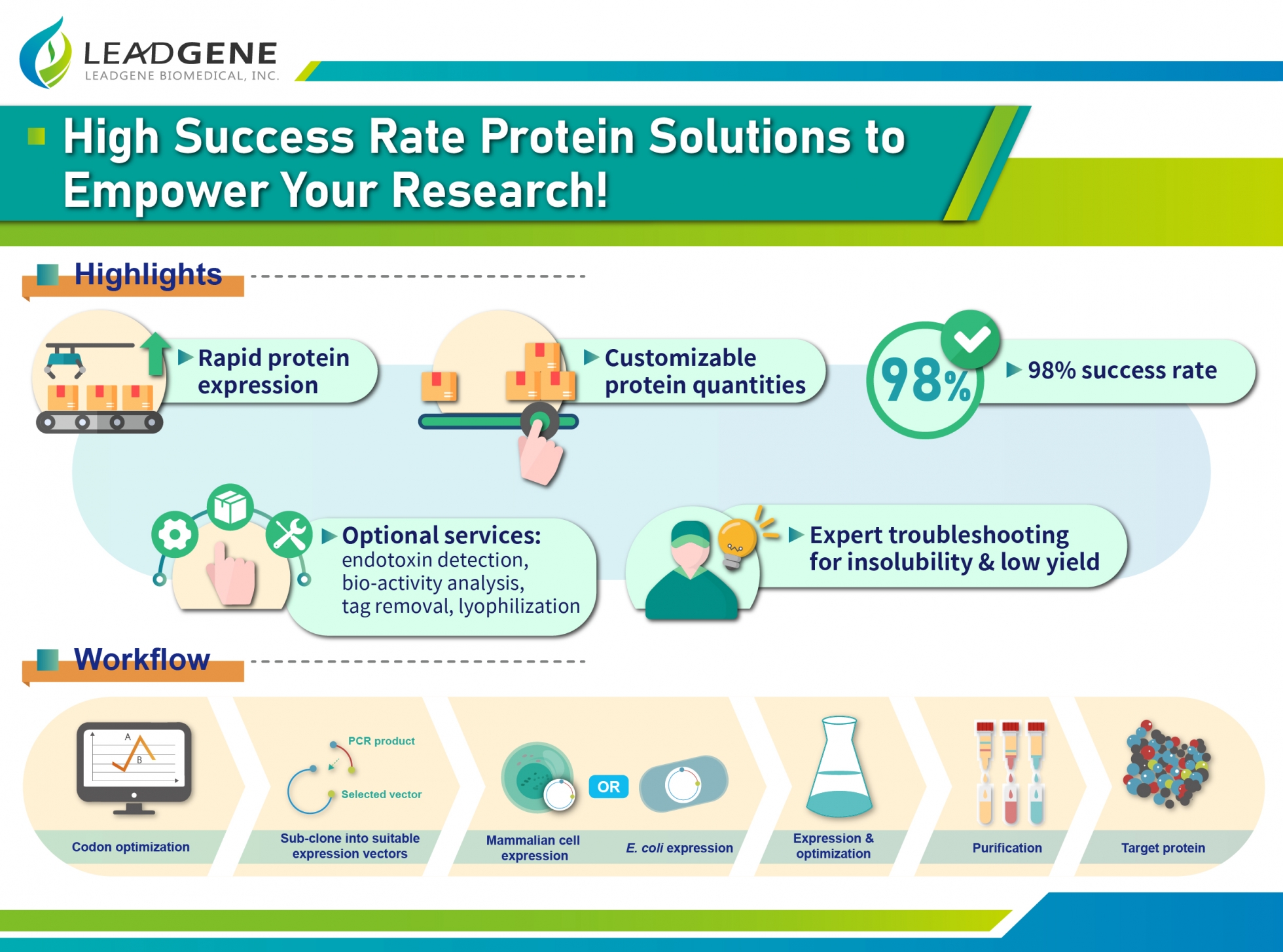
Introduction
Leadgene Biomedical is a trailblazer in the production of recombinant proteins and antibodies. We take pride in our ISO13485 and QMS (GMP) certifications, markers of our dedication to quality and compliance. We offer a wide range of services for reagent platform development, which includes creating lateral flow immunoassay (rapid test) platforms, enzyme-linked immunosorbent assay (ELISA) platforms, as well as offering services in antibody engineering, and molecular interaction analysis. As we look to the future, we are strategizing to expand our offerings to include cell line development. Leveraging our solid foundation and vast experience in the field, we provide an all-inclusive one-stop solution for the development of in vitro diagnostic reagent platforms. Our dedication to client success is manifest in our comprehensive support at every stage: from the initial design and manufacturing of raw materials to platform establishment, testing, and subsequent pilot production. Throughout this multifaceted process, Leadgene Biomedical stands alongside its clients, offering expert consulting services to ensure smooth integration and flawless application.
Features / strengths
1. An ISO13485/QMS certified plan
2. One-stop services
3. Consultative customized services
4. A comprehensive solution provider
Specification in detail