Introduction
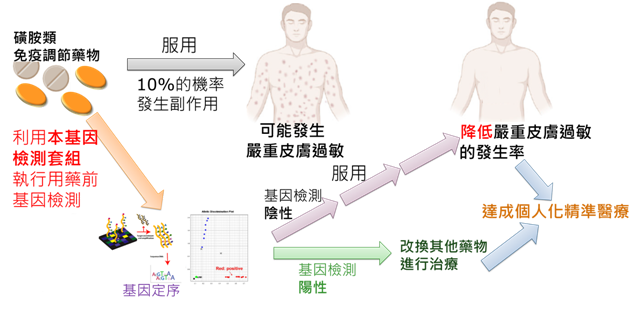
1. It has been reported that cancer patients receiving targeted therapy-EGFR inhibitors have a greater than 10% of patients may cause cutaneous adverse events. Of these, 0.5% of patients were more likely to trigger severe and fatal hypersensitivity reactions. This present invention provides a useful screening kit to detect HLA allele genes, which represents the risk of severe cutaneous adverse drug reactions induced by
novel generation of EGFR inhibitors, and the sensitivity can reach up to 81-90%.
2. Methods And Kits For Detecting Cutaneous Adverse Drug Reactions Induced By EGFR Inhibitor And Uses of The Kits: A test kit for assessing the risk of patients developing cutaneous adverse drug reactions, comprising a kit for determining specific HLA alleles and use of the test kit in assessing the risk of patients developing cutaneous adverse drug reactions.
Features / strengths
-
Specification in detail